Hydrogen embrittlement is particularly critical when using high-strength steel, and testing e.g. bolts before installation can provide a good indication of whether the bolts contain hydrogen from the manufacturing process.
Hydrogen Cracks and Hydrogen Embrittlement
Hydrogen cracks and hydrogen embrittlement are both phenomena that lead to cracks in steel materials. Almost everyone familiar with welding has at some point encountered the phenomenon of hydrogen cracks.
The phenomenon of hydrogen embrittlement is less known, but the damage mechanism is the same. Hydrogen and brint (Danish for hydrogen) is the same element, which under adverse conditions can cause cracks in steel materials. It is primarily high-strength steel and materials with locally increased hardness that are most sensitive to hydrogen cracks and hydrogen embrittlement.
Hydrogen is an element that can be present in steel just like many other alloying elements. However, the similarity ends there, as hydrogen is usually an unwanted element.
Accumulated Hydrogen Reduces Strength
Hydrogen is element number 1 in the periodic table. Hydrogen atoms are so small that they can diffuse through steel, and under certain conditions, hydrogen can become trapped/accumulated inside the steel material. Depending on the type of steel, the hydrogen that remains inside the steel can be extremely damaging to the mechanical properties of the steel.
Accumulation of hydrogen will occur along the grain boundaries in the material. After some time, cavities will form due to the internal pressure that arises. When the pressure becomes sufficiently high, the toughness and strength of the steel are reduced to such an extent that a fracture can occur. The steel becomes more brittle, and therefore it is called hydrogen embrittlement.
The mechanism is illustrated below, where the red dots represent hydrogen atoms.
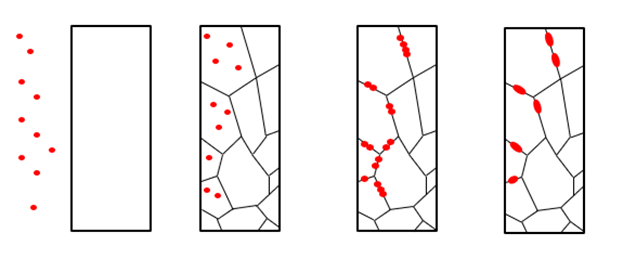
The best-known source of hydrogen in connection with welding is moisture and insufficiently cleaned base materials. The solubility of hydrogen is relatively high in molten weld metal, but the moment the weld metal solidifies, the solubility drops, and hydrogen will accumulate in the weld metal or diffuse into the base material.
Due to the heat effect from the welding, the microstructure in the fusion line and the heat-affected zone is often altered. Hydrogen will typically gather here, which can cause hydrogen cracks. Due to the stresses in the solidified weld metal, hydrogen cracks can also be seen in the transverse direction within the weld metal itself.
A few examples are shown below, where the hydrogen cracks are marked with blue arrows. Usually, the diffusion process takes time, and it is not uncommon for hydrogen cracks to appear several days after welding is completed. This also explains why many manufacturing standards require that 24 to 48 hours must pass before non-destructive testing of weld seams can be performed.

There are standards and literature that describe how hydrogen cracks can be avoided. In general, hydrogen cracks—caused by welding—are not a problem in industry today due to awareness of the issue.
Hydrogen from Corrosion Processes
In acidic environments, which can occur in crevices in a joint or from corrosion in seawater, hydrogen ions will be present. These ions react with free electrons from dissolved iron and form hydrogen gas according to this reaction: 2H+ + 2e- → H2. This hydrogen can also diffuse into the steel material, and if it is high-strength steel or cold-formed steel materials, there is a potential risk of initiation and growth of hydrogen-induced cracking.
This cracking will continue as long as there is a corrosive environment and sufficiently high stresses present. The larger the crack becomes, the greater the stresses in the steel, and thus, the crack growth increases. Once the crack reaches a certain size, it will likely result in overload fracture, as the cross-sectional area is gradually reduced as the crack grows.
Typical examples of this type of damage are high-strength bolts exposed to seawater or corrosive environments on land. An example of fracture in a high-strength bolt of class 10.9 quality is shown below.

Use of High-Strength Steel Can Increase the Risk
The general trend is an increasing use of high-strength steels to save weight and material. Sensitivity to hydrogen-induced cracking is a combination of a corrosive environment and some level of strength—and thus hardness—of the materials. If a structure is made of high-strength steel, or includes parts of high-strength steel, it is extremely important to review whether there is a risk of hydrogen-induced cracks.
Similarly, it may be necessary to check the materials to be used to ensure that the specified hardness is not exceeded. Welding will also typically create a zone with higher hardness, so it is important to focus on controlling/reducing the hardness from the process.
Hydrogen from Manufacturing Processes – Delayed Fracture
Hydrogen may also be introduced into a steel material during the actual manufacturing process—either from steelmaking of the raw material or from surface treatment of a finished component, such as hot-dip galvanizing. A typical source of hydrogen can be pickling processes, which are often used for cleaning steel parts before surface treatment.
Cleaning, e.g., in hydrochloric acid, causes significant hydrogen evolution, which diffuses into and accumulates in the steel material. To avoid the damaging effect of the accumulated hydrogen, the steel parts must be heat-treated (baked) for several hours at about 200°C. This typically forms part of the manufacturing procedure if proper quality assurance is in place.
There is no immediate problem if hydrogen has accumulated in a component as long as it is not subjected to stress. It only becomes a problem when, for example, a bolt is exposed to tensile stress. This causes hydrogen atoms to diffuse and concentrate in areas where the material is hardest or where the microstructure is deformed—typically around the bolt head or threads.
After a certain period, if the amount of hydrogen in the material combined with stress reaches a critical level, fracture may occur. This typically happens within the first 72 hours after the bolt is subjected to tensile stress. This failure mode is also known as “delayed fracture.” An example of fracture in a 10.9 bolt as a result of the above is shown on the next page.

According to ISO 15330, ‘Preload test for determining hydrogen embrittlement,’ bolts can be tested before installation.
This provides an indication of whether the bolts contain hydrogen from manufacturing and are therefore at risk of breaking after tightening.
The photos below show a tensioning test of an M48 bolt with a torque of 9500 Nm, where the bolt passed the test.

Recommendations
High-strength steel is increasingly used in industries such as wind turbine manufacturing, large infrastructure projects, and the oil industry. The use of high-strength steel can reduce material costs but increases the risk of hydrogen-induced cracking, which should be considered in the design phase.
Downloads

Reach out to our expert Henrik Bang for more infomation.



