Background
Per- and polyfluoroalkyl substances (PFAS) are a large group of synthetically manufactured chemicals with a number of very useful properties. For example, fluorinated substances typically have water-repellent surfaces, and they are also chemically very stable. These properties have meant that PFAS, since their first production in the late 1930s, have been widespread in many different products and industries. PFAS have been termed "forever chemicals" because they are extremely difficult to break down in nature. A potential way to eliminate PFAS is to break them down thermally, either by direct incineration or with the help of pyrolysis.
Potential sources of PFAS
The Danish Environmental Protection Agency mapped in 2016 the industries in Denmark that use PFAS in their production/1/. Later studies/2/ have also identified more industries that may be potential sources of PFAS pollution. Additionally, many of the products imported into Denmark are also produced by or treated with PFAS. Overall, this means that PFAS can be found almost everywhere, including in our waste and wastewater.
In waste, some of the larger potential sources of PFAS are, for example, clothing, furniture, carpets, and other textiles that are impregnated with PFAS for water and dirt repellency, electronics treated with PFAS to prevent corrosion and generally extend lifespan, as well as a multitude of other products (e.g., Teflon-coated pans and dental floss) containing various fluoropolymers.
In wastewater, some of the main sources of PFAS are industries that use PFAS or PFAS-containing products (e.g., the iron and metal industry as well as the paper and graphic industry/1/), and from washing textiles treated with PFAS. Some of the PFAS found in wastewater can also be transferred to sewage sludge, where it may also pose a problem regarding the later utilization of the sludge/3/.
There are also a number of sources that directly contribute to groundwater pollution, such as certain pesticides used in agriculture and the previous use of firefighting foam.
Measurement of PFAS in flue gases
Challenges
Currently, there are limited options for carrying out online measurements of PFAS in air or flue gas. This is among other things because PFAS are typically only present in very small amounts, far below what can be measured by direct indicating measurement methods. In addition, several of the substances are semi-volatile and can therefore exist in both particle and gas phases simultaneously, which makes it difficult to measure with online methods. Instead, PFAS must first be collected on a suitable medium over a longer period—typically several hours. This way, the PFAS present in the flue gas are trapped and concentrated to a level where they can be measured. In a laboratory, the collected PFAS are subsequently extracted from the medium, and the extraction liquid can be analyzed for PFAS.
Another and more general challenge with measuring PFAS is that currently only a limited number of substances can be analyzed for, even though there are many thousands of registered PFAS. The existing analysis options therefore so far only cover a fraction of the substances. When it comes to measuring flue gases from the thermal destruction of PFAS, this is not necessarily a major problem. Many of the more complex PFAS are expected to be broken down into smaller and more stable substances, and many of these are already included in the common analysis packages for PFAS. However, it should be mentioned that there is currently no general method in Denmark for collecting or analyzing the very small fluorinated substances in flue gases, such as tetrafluoromethane (CF4) and hexafluoroethane (C2F6), which are potent greenhouse gases with an estimated atmospheric lifetime of 10,000–50,000 years/5/. These substances have previously been identified as possible degradation products in the thermal treatment of PFAS/6/.
A previous project for the US EPA proposed a protocol for measuring these substances in connection with aluminum production/7/. Here, the measurements are suggested to be performed as 'bag sampling' or as online measurement, and the samples analyzed with either gas chromatography combined with mass spectrometry (GC-MS), Fourier transform infrared spectroscopy (FTIR), Tunable Diode Laser Absorption Spectrometry (TDLAS), or photoacoustic spectroscopy (PAS). However, several of these methods are difficult to use in field measurements. For example, it is recommended that the FTIR detector is cooled in liquid nitrogen to achieve a sufficiently low detection limit.
In Denmark, there has so far been limited focus on degradation products from the thermal destruction of PFAS, and there is still no general method for collecting or analyzing the small fluorinated substances, as there is for the larger PFAS. The USEPA is working on a method for measuring short-chain PFAS.
Existing measurement method
The only published method currently available for measuring PFAS in flue gases is "Other Test Method 45 (OTM-45)" from the USEPA/4/. This also means that OTM-45, in various adapted versions, is the method generally used in Denmark, Europe, and other parts of the world where there is interest in measuring PFAS in flue gas. The USEPA continues to work on validating and developing the method.
OTM-45 in many ways resembles the method currently used for measuring dioxins, PCBs, and other micro-pollutants in flue gas, but with additional collection steps.
A partial stream of the flue gas is exhausted isokinetically through a heated probe. The measurement gas first passes through a heated quartz filter where particles are collected. The gas then passes through a cooler, lowering the temperature to below 20°C to condense any water vapor. Next, the gas passes through a solid adsorbent consisting of XAD-2. The gas continues through three impingers, each containing about 100 ml of demineralized water. Finally, the gas passes through another solid XAD-2 adsorbent to capture any breakthrough. This setup is shown in Figure 1.
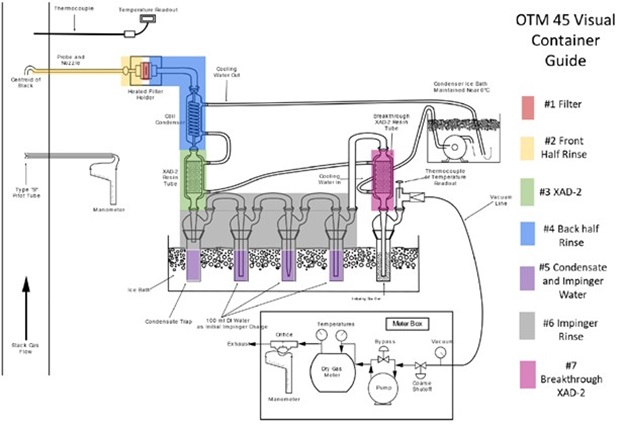
Thermal destruction of PFAS
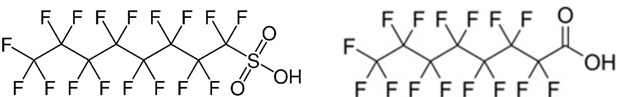
PFAS basically consist of a carbon skeleton of varying length and complexity. The carbon skeleton's hydrogen atoms are completely or partially replaced with fluorine. In addition, there may be one or more functional groups attached to the carbon skeleton, e.g., a carboxylic acid, alcohol, or sulfonic acid group. Two of the most common PFAS, perfluorooctanesulfonic acid (PFOS) and perfluorooctanoic acid (PFOA), are shown below in Figure 2.
If PFAS are completely combusted, they are converted into CO2, H2O, HF, and possibly SO2 if they contain sulfur. Whether it is possible to achieve complete combustion depends on many different factors, including the combustion temperature and residence time. The primary bond in PFAS, C-F, is a strong bond, and therefore both high combustion temperatures and/or long residence times are required to ensure complete combustion. In general, the shorter the carbon chain, the higher the temperature required to break down the specific PFAS, up to CF4 which requires about 1400°C for complete destruction/10/.
The Danish Environmental Protection Agency, in a report on the destruction of various environmental toxins/6/, reviews several articles concerning the thermal destruction of PFAS. The general conclusion in the report is that PFAS are rarely combusted completely, but instead are largely broken down into volatile fluorocarbons such as CF4 and C2F6. Currently, these substances are not measured, so their content in flue gas is unknown.
The articles reviewed in the Environmental Protection Agency’s report primarily focused on the destruction of PFOS and other simple, and typically very widespread, PFAS. This focus means that there is little knowledge about the destruction of more complex PFAS or PFAS with long carbon chains. However, it is likely that these substances will also be degraded completely or partially. There is, however, a risk that some of the more stable short-chain PFAS will be formed and then emitted with the flue gas/11/.
The thermal destruction of waste containing PFAS can roughly be divided into three different categories: ordinary incineration, where there is a surplus of oxygen; gasification, where the waste is heated with an oxygen deficit; and pyrolysis, where the waste is heated in the complete absence of oxygen. In both gasification and pyrolysis, a hydrocarbon-rich gas is formed, which can subsequently be combusted, as well as a by-product with various other applications. The distribution of PFAS in the two fractions will depend on many factors and must be investigated for the specific process and the specific waste.
Incineration
With ordinary incineration, there is a surplus of oxygen, so the degree of destruction is primarily dependent on temperature and residence time as described above. Depending on the composition of the waste and the amount and type of PFAS in it, there is a risk that some PFAS will not be completely destroyed, and thus there may be emissions of PFAS to the environment with the flue gas.
Gasification
Since the waste in gasification is heated with an oxygen deficit, there is no actual combustion of PFAS. However, some of the more complex PFAS can be expected to be broken down at temperatures above 600°C into smaller and more stable PFAS. The high temperature during gasification generally ensures that all PFAS are transferred into the gas phase and thereby into the gas that is subsequently combusted. During the subsequent combustion of the gas, a high temperature and sufficient residence time is still required to ensure that all vaporized PFAS are completely combusted. Gas engines generally do not achieve sufficiently good combustion and are not suitable. However, more modern gas turbines and gas-fired boilers are expected to ensure good combustion of the gas from the gasification process and therefore effective thermal destruction of PFAS. It is easier to achieve good combustion of gas rather than solid material, so it can be expected that complete combustion of PFAS is significantly easier to achieve with the gasification process.
Pyrolysis
Many of the same points that apply to gasification also apply to pyrolysis. However, here there is also a possibility that extra destruction of PFAS already occurs in the pyrolysis step via hydrodefluorination, as the lack of oxygen creates a reducing environment/8/. As with gasification, the subsequent combustion of the created gas is crucial to ensure the destruction of all PFAS.
The effectiveness of pyrolysis for destroying PFAS in sewage sludge has been tested at a pyrolysis plant set up at Odsherred Forsyning in Fårevejle/9/. Here, measurements in both sludge before pyrolysis, and in biochar and flue gas after pyrolysis, showed that the PFAS present in the sewage sludge could not be found in either biochar or flue gas. It was concluded that the pyrolysis ensured total destruction of the PFAS measured. Short-chain degradation products with fewer than four carbon atoms were not analyzed.
References
/1/ Nicolajsen, E.S., Tsitonaki, K., ”Kortlægning af brancher der anvender PFAS”, Miljøstyrelsen, november 2016.
/2/ Rothenborg, M., ”Disse brancher er oversete kilder til PFAS-forurening”, Ingeniøren, 4. oktober 2022, https://pro.ing.dk/watertech/artikel/disse-brancher-er-oversete-kilder-til-pfas-forurening
/3/ Ottosen, A. R., et al, ”Alternativ udnyttelse af spildevandsslam”, Miljøstyrelsen, april 2022.
/4/ ”Other Test Method 45 (OTM-45) Measurement of Selected Per- and Polyfluorinated Alkyl Substances from Stationary Sources”, USEPA, 13. januar 2021.
/5/ Marks J, et al., ”Protocol for measurement of tetrafluoromethane and hexafluoroethane from primary aluminum production”, Light Metals 2003, https://www.epa.gov/sites/default/files/2016-02/documents/tms_protocol.pdf
/6/ Geertinger, A., Jensen, A.A., Hansen, M. W., ”Belysning af destruktion af visse POP-stoffer på konventionelle affaldsforbrændingsanlæg til forbrænding af hovedsageligt ikke-farligt og for- brændingsegnet affald”, Miljøstyrelsen, juli 2019
/7/ ”Protocol for Measurement of Tetrafluoromethane (CF4) and Hexafluoroethane (C2F6) Emissions from Primary Aluminum Production”, US-EPA, Voluntary Aluminum Industry Partnership (VAIP) and The International Aluminum Institute (IAI), april 2008.
/8/ Winchell et al, ”Per- and polyfluoroalkyl substances thermal destruction at water resource recovery facilities: A state of the science review”, Water Environment Research, 93: 826–843, 2021
/9/ Wieth, C., ”White paper: PFAS elimination by Pyrolysis”, AquaGreen, 2. januar 2023.
/10/ EPA, ”Per-and Polyfluoroalkyl Substances (PFAS): Incineration to Manage PFAS Waste streams”, US EPA Tech. Brief, 2020.
/11/ Meegoda, Jay N., ”A Review of PFAS Destruction Technologies”, International Journal of Environmental Research and Public Health, december 2022.
Downloads

Reach out to Lars Piilmann Brorholt for more information.



