Good hygiene is an absolute necessity for producing safe food. Therefore, cleaning conditions and validation are crucial factors in safe business operations and production.
In a modern production company, many cleaning operations are automated, making the process faster, less manpower-intensive, and reproducible. Closed production equipment typically uses Cleaning In Place (CIP) because manual cleaning is not possible. The concept of Cleaning In Place was introduced back in the 1950s and is now an integral part of the cleaning process in all major food and pharmaceutical companies. As cleaning is conducted automatically in usually closed systems, it is important to have a reliable method for validating that the cleaning is sufficiently effective.
Method for validating effective CIP cleaning
The common method for validating effective CIP cleaning in newly installed production equipment typically involves:
- covering the surfaces in the equipment, i.e., tanks and pipes, with a fluorescent test liquid,
- cleaning according to standard procedures,
- examining the equipment with a UV source,
- observing the result: where residues of the test liquid remain, the cleaning is not sufficient.
For many years, riboflavin suspended in water has been the 'standardized' and recommended fluorescent test liquid for this type of cleaning validation (described by VDMA, German Process Plant and Equipment Association1). But is the riboflavin solution representative enough of the products passing through the production equipment, considering viscosity and adhesiveness? And does riboflavin reveal what you are looking for?
This has been the starting point for a series of experiments conducted by FORCE Technology in collaboration with Alfa Laval at Alfa Laval's pilot plant. Here, the effectiveness of cleaning has been examined using both riboflavin and an alternative fluorescent test fluid on metal plates, pipes, and in a tank.
Alternative to riboflavin
An alternative fluorescent penetrant (FP-10, Magnaflux) was tested instead of riboflavin. Penetrant is known and used in penetrant testing—an NDT (non-destructive testing) method used to detect microscopic cracks and crevices open to the surface. The penetrant adheres well to metal surfaces and penetrates cracks and other openings via capillary action. By combining the penetrant with a developer, you can locate defects in surfaces not visible to the naked eye.
Experiments with metal plates
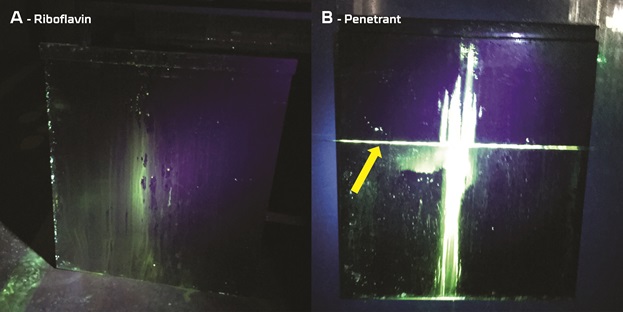
In a series of experiments, 1x1 m metal plates were covered with either riboflavin or penetrant. The plates were then hung up for cleaning by a CIP nozzle for 2 minutes. To simulate a blind cleaning angle, a pipe was placed in front of the plate, casting a shadow on the water jet from the CIP nozzle (see Figure 1). After washing, the plates were illuminated with a UV source to detect any remaining riboflavin or penetrant, revealing the blind cleaning angle.
The experiments showed that riboflavin is very easily washed off metal surfaces and that the pipe simulating a blind cleaning angle is not visible after washing (see Figure 2 – Image A). In contrast, both the pipe and the string that cast a shadow during cleaning become clearly visible in experiments using penetrant (see Figure 2 – Image B).

Same experiments conducted in a tank
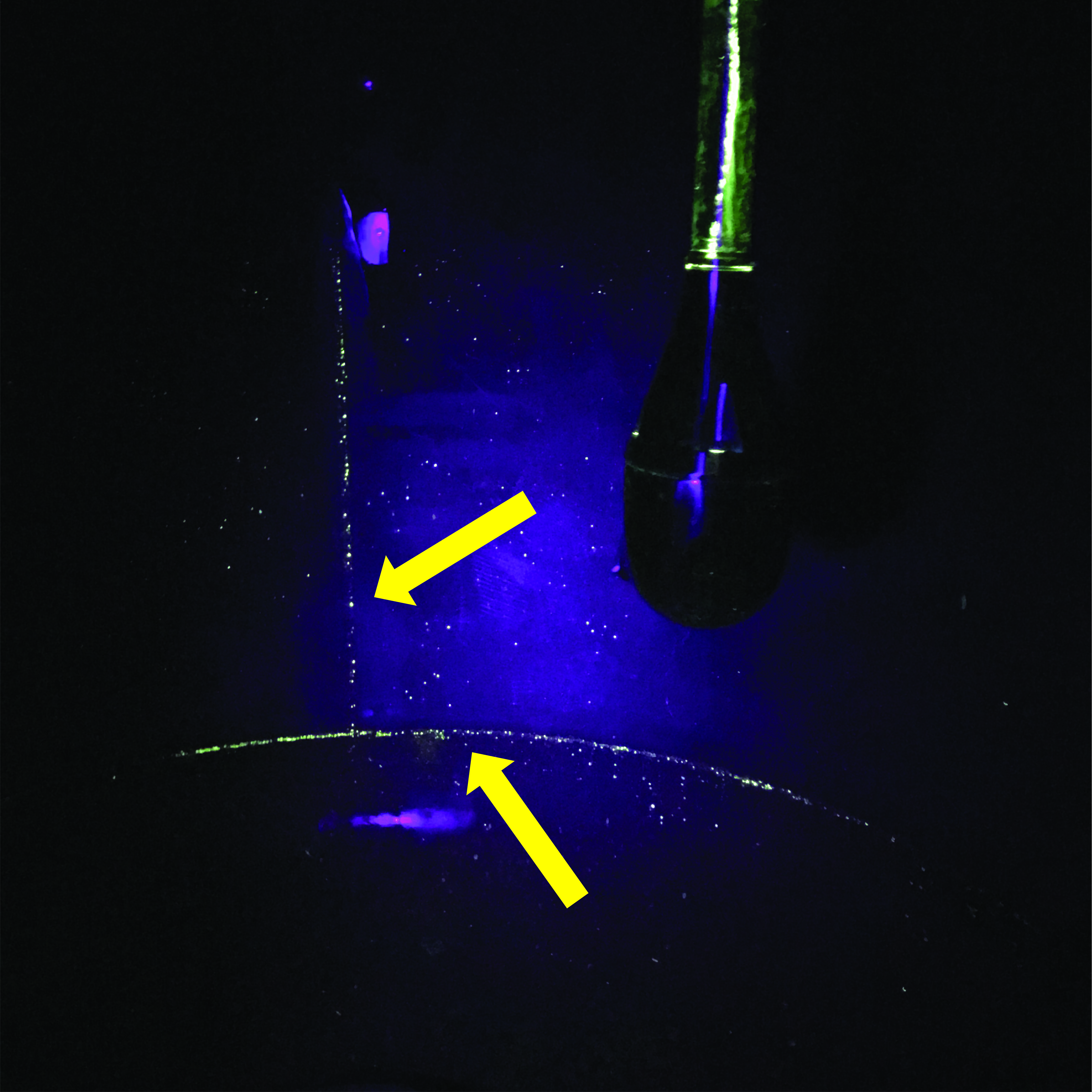
The experiments were subsequently repeated in a tank, where cleaning is also performed by a CIP nozzle for 2 minutes. Again, the tank's surface was covered with either riboflavin or penetrant. In general, riboflavin's ability to adhere to the tank surface before washing is much poorer than that of the penetrant (see Figure 3 – Image A, B, and C). The penetrant spreads uniformly over the surface, whereas riboflavin "beads up" and runs off.
On the glass door of the tank, riboflavin is easily washed off and disappears after the first water jet hits the door. After washing, both the glass door and the tank walls are completely clean of riboflavin. In contrast, the penetrant requires several washes to be removed from the glass door. After washing, the pipe to which the CIP nozzle is mounted is not completely clean. Furthermore, the penetrant also reveals welds that have not been ground and polished on the tank's surface (see Figure 4).

Experiment performed in pipes
The penetrant’s ability to validate cleaning in pipes was also tested. Using an endoscope with a UV source, it is easy to find areas in the pipe where the penetrant has not been washed away. Thus, it is possible to visualize both "dead ends" and seals where organic material can deposit and where adequate cleaning is more difficult (see Figure 5).
Riboflavin versus penetrant
Cleaning validation with fluorescent test fluids and UV lighting is usually performed before new equipment or a new production process is put into use. The purpose is to ensure that CIP cleaning is sufficiently effective. The method can also be used to locate sources of contamination if recurring cleaning failures are observed.
The experiments comparing riboflavin and penetrant show that riboflavin is very easily washed off surfaces, and in some cases, aerosols alone are sufficient to remove it. This easy removal, however, may be detrimental as blind cleaning angles—often sources of contamination—will not be visualized.
Additionally, riboflavin often does not represent the medium adhering to surfaces during and after production due to its poor adhesion. Therefore, the cleaning's effectiveness may be overestimated when using riboflavin, potentially approving cleaning validation on a false basis.
As an alternative to riboflavin, FORCE Technology recommends using penetrant, which has a far better ability to spread and adhere to metal surfaces. The penetrant is harder to wash off and thus provides a more indicative result of whether equipment has been efficiently cleaned or not. As an added benefit, cracks, crevices, and corrosion holes are easier to spot, especially if the penetrant is used in combination with a developer.
There are several penetrants that are NSF-registered2 and therefore recommended for food-contact surfaces. After the experiments were completed, the penetrant used here (FP-10) was discontinued, but other suitable alternatives tested by FORCE Technology are available and also NSF-registered. For more information, contact Kristine Garde.
Notes:
1. Information sheet Riboflavin test for low-germ or sterile process technologies Fluorescence test for examination of cleanability For food, aseptic, pharmacy and chemistry, VDMA 2007
2. Nonfood compounds and proprietary substances found compliant to food safety regulations are listed in the NSF White Book™
The article was written by:
Kristine Garde, Annette Baltzer Larsen and Palle Korning Ove, FORCE Technology & Kim Kjellberg, Alfa Laval
The article was previously published in Plus Proces No. 3 - 2019
Downloads
Experiences applied in new hygiene project
As mentioned in an article in Plus Proces no. 2 2019, a new project with the focal point of "hygiene" has been launched at FORCE Technology. The project is based on two tracks: in one track, sensors are used to assess cleanliness, while in the other track, methods are being developed for condition assessment of the equipment’s surfaces.
It is specifically in the development of new methods for condition assessment of equipment surfaces that FORCE Technology’s experience with fluorescent liquids is applied. This ensures that the most suitable fluorescent liquid is chosen for further work in the development of a new evaluation tool, the “Scoreatlas.”
The Scoreatlas is a method for objective visual evaluation of a stainless steel surface’s condition, independent of the assessor’s professional background and experience. With a Scoreatlas, you can monitor the development/condition of the equipment over time and decide when it is necessary to repair damages, depending on the requirements imposed on each producer.
The project is co-financed by the Ministry of Higher Education and Science/The Agency for Institutions and Educational Grants. For further information about the project, contact Annette Baltzer Larsen.

Reach out to Annette Baltzer Larsen for more information.



