By Annette Baltzer Larsen
It is generally known that production lines must be clean and cleaning processes optimised. But compared to what, and most importantly, how do you best measure this?
There are differences in optimised cleaning processes
It can be challenging to determine how best to validate the cleaning of your production equipment—that is, to check whether the cleaning has been optimised. How do you ensure that potential shortcomings are detected in time?
There is also the question of whether the chosen cleaning procedure can be adapted to the specific cleaning process desired, as well as whether the cleaning validation is organised in such a way that the most hygienically critical areas are examined.
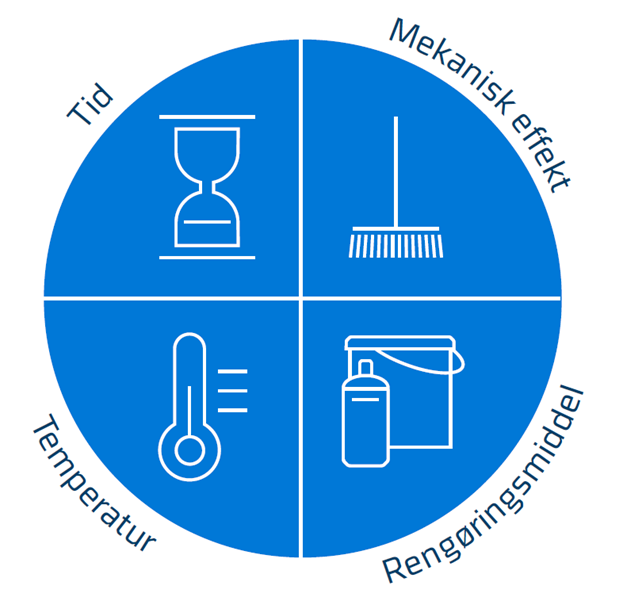
An optimised cleaning process for one production unit is not necessarily optimal for another. When considering the four parameters important in the cleaning process, it is essential that they all fill the "pie chart" (see Figure 1), which indicates an optimised cleaning result where all biofilm formed during production has been removed.
The Sinner’s Circle ensures an optimised cleaning procedure
Optimised cleaning should, figuratively speaking, fill a pie—the so-called Sinner’s Circle, see Figure 1. The cleaning process used should be optimised to fit the specific production—such as production type, duration, type of organic material soiling the surfaces, and the hygienic design of the production equipment.
As shown in Figure 1, the four parameters involved in the cleaning process are:
- The cleaning agent, which should be chosen according to the type of soiling (organic matter) present on/in the production equipment.
- The cleaning temperature, which should be adjusted to the temperature at which the cleaning agent works best, but also to the organic material to be removed.
- The cleaning time, which should be long enough to remove the biofilm built up during production time.
- Mechanical action is the most difficult parameter to optimise throughout the production equipment. Cracks, crevices, and sharp angles may be hard to access in open systems and difficult to achieve good flow in closed systems.
Let’s take, for example, a pan from last night's Bolognese: soaking in cold water is not very effective. Nor is adding a little dish soap to the cold water. However, using hot water with soap improves cleaning somewhat, but it only becomes really effective when mechanical action from a scrubbing brush is also applied. In other words, all four parameters in the Sinner's Circle must be used.
The distribution of the four parameters in the "pie" will vary depending on the cleaning procedure chosen.
Regardless of the cleaning procedure, the equipment used must also be constructed according to hygienic principles. That is, CIP and COP systems must be hygienically designed with internal cleaning systems to keep themselves clean. The cleaning solution must reach all surfaces to be cleaned, and temperature and cleaning agent concentration must be maintained at set-points (that is, the temperature and concentration in the return flow).
For manual cleaning, brushes, scrapers, foamers/low-pressure systems, and so on, must also be kept clean.
All of this is crucial to prevent cross-contamination—carryover of microorganisms or allergens from one production cycle to the cleaning fluid/equipment and then back to the production equipment.
Risk-based approach to the cleaning process
Regardless of which cleaning procedure is chosen for the specific cleaning process, it is important to take a risk-based approach to cleaning.
The cleaning fluid must reach all areas of the equipment to keep it clean. With a risk-based approach, the equipment should therefore be inspected, ideally both during production and during cleaning. It is key to identify hygienically critical hotspots so that extra attention can be paid to these spots during cleaning and so that they are included in cleaning validation. These critical hotspots are often places where product accumulates during production and where thorough cleaning is particularly challenging.
Common cleaning procedures in the food industry
According to 3-A Sanitary Standards Inc., cleaning procedures within the food and pharma industries are typically divided into four main types—some well known, others only used in parts of the industry.
1. Cleaning In Place (CIP)
CIP means cleaning the complete production equipment without disassembling or opening it—only minimal (or no) manual operation by the user is needed. There must be a CIP pump to move the cleaning fluid to the objects at the appropriate flow.
The mechanical effect on surfaces occurs by flow or pressure as the cleaning fluid circulates through the production equipment.
CIP is used for closed production equipment such as pipes, tanks, and pumps.
Key points to remember
Temperature and cleaning fluid concentration are only measured at the return flow, and the clock only starts counting once those have reached the set-point—i.e. when temperature and concentration are optimal for the specific cleaning job. Time must be long enough for all biofilm built up to be washed off during cleaning.
The most difficult parameter to control in the CIP process is mechanical action: how is the hygienic design of the production line being cleaned? Are there dead ends, cracks, sharp angles, or pitting, where cleaning fluid struggles to reach with sufficient flow? Cleaning will be less effective in these areas.
Water from previous final rinses is often collected and reused for the next pre-rinse. This can be problematic if cleaning is not optimised, as microorganisms and allergens can be present in this water. It is therefore essential to monitor the quality of pre-rinse water to assess whether the final rinse water is clean enough.
2. Cleaning Out of Place (COP)
In COP cleaning, sub-equipment is removed and placed in a dedicated cleaning station—like a tank with recirculating cleaning fluid or a dishwasher. This is thus a partially automated process, with the operator dismantling and placing equipment in the cleaning station. Mechanical action on surfaces comes from the flow or pressure of the cleaning fluid recirculating in the station.
For some sub-equipment, cleaning effectiveness can depend considerably on whether it stands or lies in the station, because the cleaning fluid must reach all surfaces with appropriate mechanical action. After washing, sub-equipment is remounted in the production line.
Key points to remember
The temperature and concentration of cleaning agent must be high enough before the cleaning process countdown begins. If the set-points are not reached when cleaning starts, part of the cleaning will not be optimal.
Mechanical effect can vary greatly depending on the cleaning station used; some only circulate fluid with little flow, or perhaps with too high a flow, which can be equally poor. In some stations, cleaning fluid might not even reach the farthest areas. Others distribute fluid more effectively, with mechanical action adapted to the task. The positioning of equipment in the station can be vital for fluid flow to be an effective parameter in the process.
As in CIP cleaning, the reuse and quality of water must also be evaluated.
3. Manual Cleaning
A large part of the operator’s/sanitation worker’s time is spent on manual cleaning. Most often foamers or low-pressure systems are used, and brushes and squeegees of different kinds for particularly critical spots that are hard to reach. Manual cleaning may require partial or full disassembly of equipment to access critical areas.
Key points to remember
Temperature and cleaning agent concentration in manual cleaning are lower than in the more closed, automated cleaning (CIP or COP). Therefore, this procedure requires more time. The mechanical effect comes from the low-pressure system/foamer and/or the brush used.
Be aware that this procedure carries increased risk for the formation of aerosols, as organic material, microorganisms/allergens, and chemical droplets may be released into the air and contaminate surfaces.
4. Dry Cleaning
Dry cleaning is used in dry productions (powder, flour, etc.), where water is undesirable. It typically uses vacuum cleaners and dry brushes for sweeping down.
Important to remember
Dry cleaning must be dry. Even a slight bit of moisture can cause problems in subsequent production. If water cleaning is occasionally necessary, rooms and equipment should be allowed to dry completely before resuming production. Full drying also applies to cracks, crevices, and pits in equipment.
Check whether your cleaning process is optimised
Is your cleaning effective enough for your specific production? Always assess whether your cleaning meets production requirements and consider how you can improve your cleaning validation.

Reach out to Annette Baltzer Larsen for more information.



