It is not an easy task to validate and verify the cleaning procedure. Here, we present methods for cleaning validation, which can subsequently be used as verification of the effectiveness of the cleaning.
Legally, products that could be potentially harmful to customers’ health must not be sold. Therefore, it is important to eliminate risks of product contamination. Contaminants can, for example, be:
- Physical contamination: Plastic or metal fragments from e.g. equipment
- Chemical contamination: Residues of cleaning agents, for example due to poor hygienic design of production equipment (cracks, crevices, dead ends, lack of drainage)
- Biological contamination: Microorganisms, formed toxins or allergens, mainly resulting from inadequate cleaning but can also be due to poor equipment construction
What is good enough for your production must be determined by a risk assessment that weighs the risks of contamination against the severity and frequency of the given risk.
In this article, our focus is on methods for cleaning validation, which can subsequently be used to verify the effectiveness of the cleaning. Once cleaning has been validated, the procedure is applied routinely, after which it is monitored and verified whether the procedure continues to be sufficient. A new validation should be carried out when there are changes in equipment, processes, or procedures, but validation should also be renewed regularly, for example after ½ - 1 year of production, or whenever something is found to be out of control.
It is important to point out that an absence of contaminants does not necessarily mean effective cleaning, but can also result from an insufficiently considered risk assessment regarding either sampling or analysis.
Methods for cleaning control – what are the options?
An essential element in validating a cleaning procedure is the risk assessment of what is clean enough? And how should “clean enough” be checked?
First step should always be to use the senses. Is the equipment visually clean, even in the places that are harder to access during cleaning, and does it smell clean? Or is there a suspicious odor, which, for example, could result from accumulated product in a less accessible place—maybe even inside the equipment itself?
If the equipment is not visually clean, or if there is a suspicious odor indicating that cleaning has not been sufficient, it is a waste of resources to take samples to check whether the cleaning was sufficient. See figure 1, where an agitator in a tank is not visually clean. In such cases, it makes no sense to use resources taking samples to follow up on whether the production equipment is clean—it is not.

Second step is then to take samples for cleaning validation. In this step, it is important to assess where the most hygienically critical places are. These are the areas that are hardest to keep clean and/or might have product accumulation during production. Consequently, these locations should be included in cleaning validation to examine whether the cleaning procedure can keep these hygienically critical sites clean and thus fulfill the set requirements even in the hardest to clean areas. In addition, it must be decided which analytical methods and thresholds are to be applied to the specific production.
Direct sampling – usually for open equipment
During direct sampling, a swab, sterile cloth, or direct print is used on the surface. The number of microorganisms or other contaminant is indicated per examined area, e.g., cm2.
The disadvantage of these samples includes the uncertainty of whether the chosen site is truly the most critical and whether it has been sufficiently swabbed to include any remaining dirt/biofilm in the sample.
In a project in collaboration with Danish Food Innovation (DFI), direct sampling is being investigated with respect to a new measuring device from SBT Instruments. Results from this study will be presented in a later article.
Indirect sampling – usually for closed production equipment
In indirect sampling, a quantity of the final rinse water is taken and then analyzed for microorganisms or other contaminants. In this way, a larger surface area of the equipment is examined. It must, of course, be ensured that the rinse water has sufficient flow for mechanical processing of equipment surfaces throughout the closed production equipment. If the flow is inadequate, there is a risk that the sample does not represent cleaning effectiveness correctly.
It is also important to remember the dilution effect when taking an indirect sample. If the dilution effect is large, it is crucial to choose the analytical method carefully to ensure meaningful results; that is, something can actually be detected in the sample even if there is significant dilution. If the result is always below the detection threshold, it should be risk assessed whether the analytical method chosen is correct for the purpose.
The choice between direct and indirect sampling depends on the production line. It is more difficult to take a direct sample that is also representative for an entire closed production line (pipes, tanks, pumps, etc.). Therefore, indirect sampling is more common for closed production equipment, whereas direct sampling is used for open equipment.
In the project “Monitoring of hygiene in closed production equipment”—funded by the Agency for Innovation and Development—we are working on various possibilities for monitoring hygiene in closed production equipment such as tanks, pipes, and pumps. To measure hygiene in the closed equipment, rinse water is analyzed, making it an indirect sample. In the project, two new cleaning validation methods are being tested and validated, described below, as well as common microbiological validation tests so the new methods can be validated against known standards. Results will be presented in a future article in Plus Proces (no. 6).
Choice of analysis
Microorganisms from a biofilm remaining on equipment after cleaning may be in a “viable-but-not-culturable” state. This means they can grow in the actual product and cause issues with food safety and product quality, but they often cannot grow or be counted using standard tests for either total count or the specific microorganism.
If microbiological cultivation and enumeration is desired, the challenge with both direct and indirect samples is that stressed microorganisms often cannot grow in the standard test. These microorganisms often need to be "nursed" or destressed before they can grow and be counted.
Chemical analyses of final rinse water generally do not pose the same challenges as microbiological counts, as chemical components are measured rather than viable microorganisms.
To measure bacteria or total organic carbon (TOC) in rinse water, new measurement methods have been introduced. The TOC analysis measures a chemical component (carbon), while bacteria measurement uses a new flow cytometry technology to count individual bacterial cells. This means there is only counting, not cultivation and counting, eliminating the problem of stressed microorganisms that do not grow in standard tests (“viable-but-not-culturable”). By skipping cultivation, a much faster measurement is obtained, and many more bacteria can be counted than with conventional methods. However, it is challenging to correlate these results to established value ranges for microbiological analyses (cultivation and counting) simply because the traditional methods detected far too few microorganisms—too many could not grow. The technology can also count particles similar in size to bacteria.
SBT Instruments – BactoBox
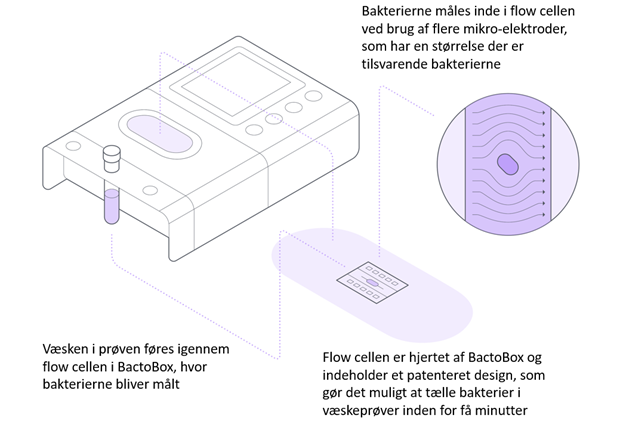
BactoBox is a completely new instrument that uses Danish-developed technology to measure the number of bacteria in liquids in just a few minutes. The technology is based on flow cytometry, well known from advanced research laboratories. The measurement principle for BactoBox is shown in figure 2. BactoBox features a flow cell with a set of micro-electrodes the same size as bacteria. When a bacterium passes over the electrodes, it is detected by the micro-electrodes. Particles are also detected and counted separately. Thus, BactoBox can detect individual bacteria before they are cultivated and quickly provide a result on the number of bacteria in the sample, with no sample pretreatment required.
Since the technology measures individual cells without cultivation, BactoBox counts all bacteria, regardless of whether they are stressed, in spore form, or have special growth conditions. Due to the “viable-but-not-culturable” challenge, it is not at all certain these bacteria will be seen if cultivated on an agar plate (standard test), but they can still cause food safety and quality problems. This also means you will typically see a higher bacterial count with BactoBox than with traditional culture and counting methods. A higher result from a BactoBox measurement is not an indication that cleaning is insufficient, but rather that the new method is more sensitive than the old and enables earlier detection if there is a trend shift.
Insatech – Total organic carbon
For cleaning control of particularly difficult production equipment or when switching between critical products, Total Organic Carbon (TOC) monitoring can be used. The TOC measurement principle is based on measuring organic carbon. Carbon is the chemical building block in almost all materials, from process products (fat, protein, microorganisms) to some of the cleaning agents used. The TOC level indicates the presence of organic material to a greater or lesser degree. Whether it originates from residual dirt, biofilm, or organic cleaning agents after cleaning, it can be measured. By comparing the result from clean water with the measurement of the final rinse water, even small differences in organic carbon content can be detected, down to ppb level.
By measuring the carbon content in the final rinse water after cleaning, a value for TOC in the rinse water is obtained. Thus, it can be assessed whether the cleaning procedure has been sufficiently effective in removing product residues or other organic material, and this information can be used to release the production equipment for the next batch. Sievers TOC analyzer is available for both laboratory use and online monitoring (see figure 3).

The correct choice of analysis and a good risk assessment of equipment, production, and processes must ensure that the thresholds for cleaning are sufficiently low to guarantee that product quality is maintained until the expiry date, and that food safety is always top notch.
When choosing an analytical method, it is important to assess the lower detection limit for the specific analysis—is it good enough in terms of the risk assessment made for production and equipment?
The subsequent assessment of monitored results is made using the thresholds defined in the risk assessment and validation process. Verification of measurements should continue until a new cleaning validation is performed.
When is cleaning sufficient?
A comprehensive risk assessment of equipment, production, and processes must underpin when cleaning for a given production is sufficient. This must naturally be combined with shelf-life testing to ensure food safety and desired product quality at the set expiry. It is important to choose analytical methods that provide a real assessment of cleaning effectiveness. If results are always below the detection limit for cleaning validation, it should be reviewed whether the chosen analysis is appropriate or if the sample taken is representative for the production line. Setting the standard for when cleaning is sufficient is a significant task. However, it becomes easier if methods that are both fast and sufficiently sensitive are used—even when a low detection limit is required.
In the project, both new methods are used alongside standard microbiological methods in two case companies from different branches of the food industry. Exciting results have emerged from the first case company, and the first half of the results from the second company also look promising. The next article will present key results from case company 1.
This article was first published in Plus Proces in August 2020.

Reach out to Annette Baltzer Larsen for more information.



